|
4/2/2023 0 Comments Standard entropy change  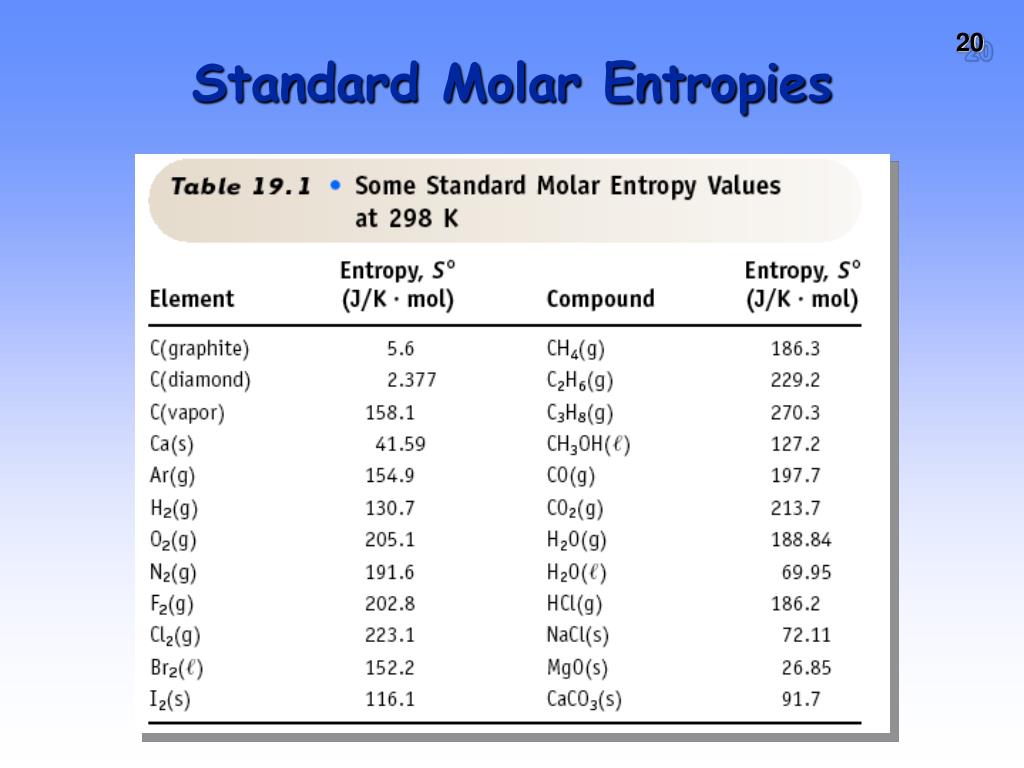
NaCl, MgCl₂) and relevant energy terms for: the construction of Born–Haber cycles and related calculations b) use of the lattice enthalpy of a simple ionic solid (e.g.a) explanation of the term lattice enthalpy (formation of 1 mol of ionic lattice from gaseous ions, ΔLEH) and use as a measure of the strength of ionic bonding in a giant ionic lattice.Module 5: Physical chemistry and transition elements.be able to use the equation ΔG = ΔH − TΔSsystem to predict whether a reaction is feasible, and to determine the temperature at which a reaction is feasible know that the balance between the entropy change and the enthalpy change determines the feasibility of a reaction and is represented by the equation ΔG = ΔH − TΔSsystem be able to calculate the entropy change for the system, ΔSsystem, in a reaction, given the entropies of the reactants and products understand why entropy changes occur during: changes of state dissolving of a solid ionic lattice reactions in which there is a change in the number of moles from reactants to products know that entropy is a measure of the disorder of a system and that the natural direction of change is increasing total entropy (positive entropy change) understand the effect of ionic charge and ionic radius on the values of: lattice energy enthalpy change of hydration be able to use energy cycles and energy level diagrams to carry out calculations involving enthalpy change of solution, enthalpy change of hydration and lattice energy be able to define the terms ‘enthalpy change of solution, ΔsolH’, and ‘enthalpy change of hydration, ΔhydH’ be able to construct Born-Haber cycles and carry out related calculations be able to define lattice energy as the energy change when one mole of an ionic solid is formed from its gaseous ions Perform calculations of an enthalpy change using these cycles.Students should be able to: define the term enthalpy of hydration.Cycles are used to calculate enthalpies of solution for ionic compounds from lattice enthalpies and enthalpies of hydration.Construct Born–Haber cycles to calculate lattice enthalpies using these enthalpy changes.Born–Haber cycles are used to calculate lattice enthalpies using the following data: enthalpy of formation, ionisation energy, enthalpy of atomisation, bond enthalpy, electron affinity.Lattice enthalpy can be defined as either enthalpy of lattice dissociation or enthalpy of lattice formation.Use the relationship ΔG = ΔH – TΔS to determine the temperature at which a reaction becomes feasible.Use the relationship ΔG = ΔH – TΔS to determine how ΔG varies with temperature.Students should be able to: calculate entropy changes from absolute entropy values.ΔS accounts for the above deficiency, illustrated by physical changes and chemical changes.The concept of increasing disorder (entropy change, ΔS).Gibbs free-energy change, ΔG, and entropy change, ΔS.RSC Yusuf Hamied Inspirational Science Programme.Introductory maths for higher education.The physics of restoration and conservation.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
